 Ne Mg Cl Si 10 P 10 N 18 N 14 N 12 P 12 N. 
Place dots around the element’s symbol one at a time (can’t exceed 8). Figure out how many valence electrons the element has in its atom. The 1st energy level can hold up to 2 electrons. Draw Atomic models using the Bohr Model configuration Calculate the number of protons, neutrons, and electrons Determine Atomic Number, Atomic Mass, Number of Protons, Number of Electrons, and Number of Neutrons Draw Nucleus and Electron Orbitals Templates to identify:Group, Period, Protons, Neutrons, Electrons,Energy Levels, Valence Electrons Draw electrons and input proton and neutrons in nucleus Based on Common Core: NGSS: MS-PS1-5, MS-PS1-1 CCSS:RST.11-12.2, RST.11-12.4, RST.11-12.6, RST.11-12.7, RST.11-12.10, NGSS:HS-PS1-1, HS-LS1-6, HS-PS1-2 Ink and Paper Type: Black & white interior with white paper Bleed Settings: Bleed (PDF only) Paperback cover finish: Matte Trim Size: 8. CREATING BOHR DIAGRAMS Rules for arranging electrons: 1. Atomic Structure Models Bohr Models Practice Templates This Templates simplify the creation of atomic structures The circles for circular orbits of the electrons Dot represents the hydrogen atom nucleus Identify Bohr Models,Draw Bohr Models Bohr Model Atoms Review Templates Worksheets Word search puzzles and Cryptograms with words hidden in puzzle worksheet. Electrons can jump from one orbit to another by emitting or absorbing energy. Starts with puzzles based on Bohr model of the atom. The orbits are labeled by an integer, the quantum number n. In the Bohr model of the atom, electrons travel in defined circular orbits around the nucleus. Showing top 8 worksheets in the category - Bohr And Lewis Dot Model. Other contents: physics Share / Print Worksheet. Many reaction intermediates with variable. How many electrons can each shell hold a. Before fixing the sheet revise the whole chapter from your textbook and ensure you have revised the physics method used within the chapter. Bohr Model Worksheet Use the description sheet and the periodic table to help you complete the following Bohr models. School subject: Physics (1061802) Main content: Atomic (2002159) Bohr model. It consists of a dense core called the atomic nucleus surrounded by a space occupied by an electron cloud. They are also all listed on TPT if you prefer buying that way.COMMON CORE BOHR MODEL PRACTICE WORKSHEET: LEARN ATOMIC STRUCTURE BOHR ATOMIC MODEL TEMPLATES PRACTICE WORKBOOK FOR CHEMISTRY SUCCESS Research has found that repetition is an essential learning aid. Liveworksheets transforms your traditional printable worksheets into self-correcting interactive exercises that the students can do online and send to the teacher. It goes great with the Periodic Table Battleship game we created, too! Also check out this post on how to Teach Chemistry to Kids. The printable cards include the following elements: We loved this fun interactive chemistry lesson! Physics (1061802) Main content: The Bohr Atomic Model (1747417) The Bohr Atomic Model. When you have multiple rings for the orbits, make some a little smaller than others so you can layer them. Liveworksheets transforms your traditional printable worksheets into self-correcting interactive exercises that the students can do online and send to the teacher. To represent the of protons write a P- followed by the number of protons. Just add the needed number of beads onto the pipe cleaner and twist it together. Draw Bohr atomic models for each of the atoms using your Periodic Table 2. We used Pipe Cleaners for the orbits, Pony Beads for the electrons and Pom Poms for the nucleus of our atomic models. The atomic number of the elements represents the number of electrons the element has. In this study, this consideration is translated by preparing a tutorial worksheet for Bohrs Atomic Model according to the suggestion of three learning levels of chemistry. Which two statements are correct about the Bohrs atomic model Select two (2) boxes. What is the difference between the atomic number & the mass number of an element 3. 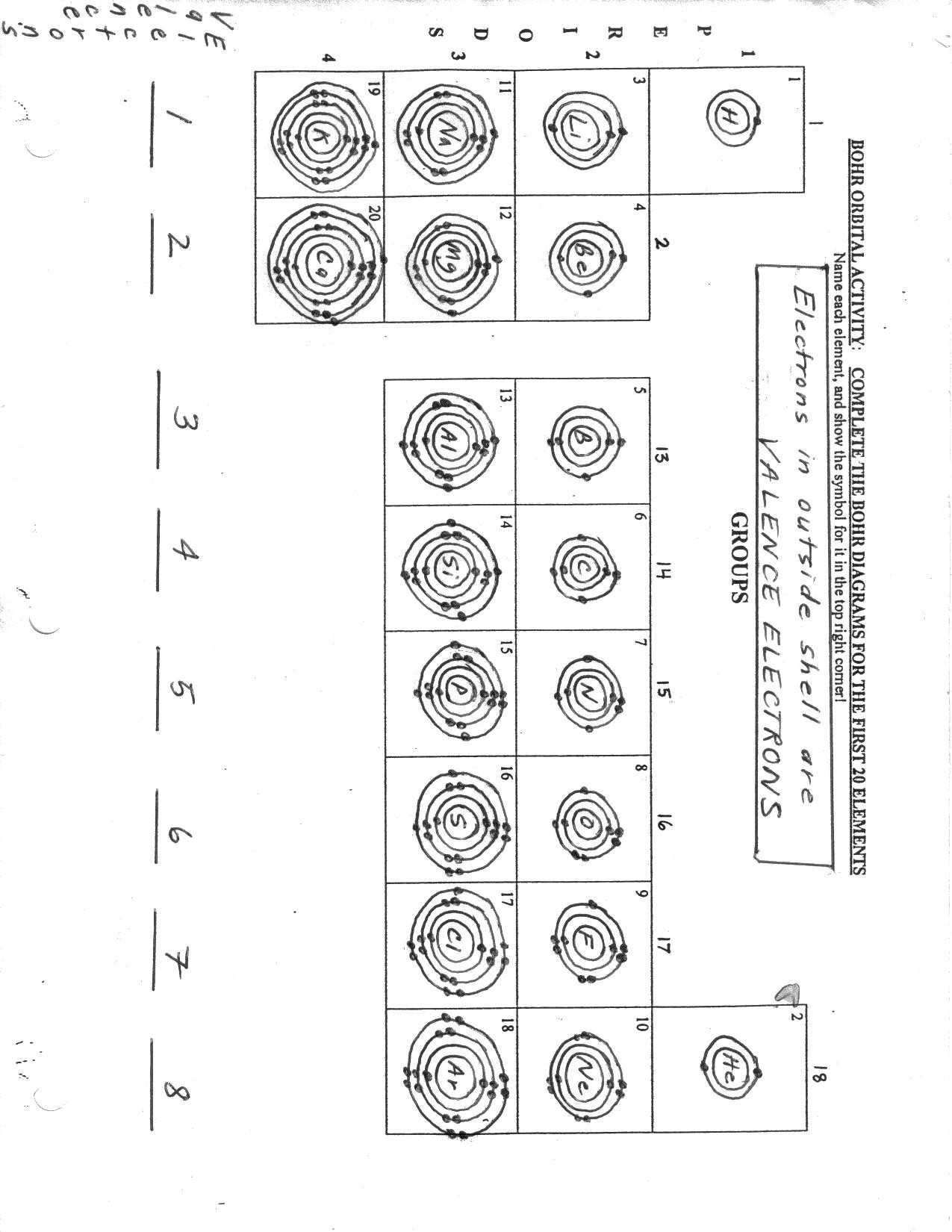
Thomson’s plum pudding model, Rutherford’s planetary model, and Bohr’s electron shell model in order to highlight pivotal changes in our. What two sub-atomic particles are located in the nucleus of the atom 2. The Bohr Atomic model has a positively charged atomic nucleus with negatively charged electrons circling the nucleus on orbits (the circular lines). If your teacher asks you to pause the video and look at the worksheet you should: Click 'Close Video' Click 'Next' to view the activity Your video will re-appear on the next page, and will stay paused in the right place. describe different models of the atom in order to explain how the atomic model has changed over time, recall the details of Dalton’s hard sphere model, J. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
